168飞艇官网开奖结果记录直播 幸运飞行艇官方开奖记录查询结果-开奖历史记录查询 幸运168飞开奖艇官网开奖历史 Stop attacks on your cloud native applications
We prevent them before they happen,
stop them as they happen
Get a Demo
Unify cloud security to detect, prioritize, and reduce risk
See and stop threats across every phase of your software development lifecycle,
from code to cloud and back
from code to cloud and back
Dev Security
Secure your weakest software supply chain links and prevent security risks from becoming security incidents
Cloud Security
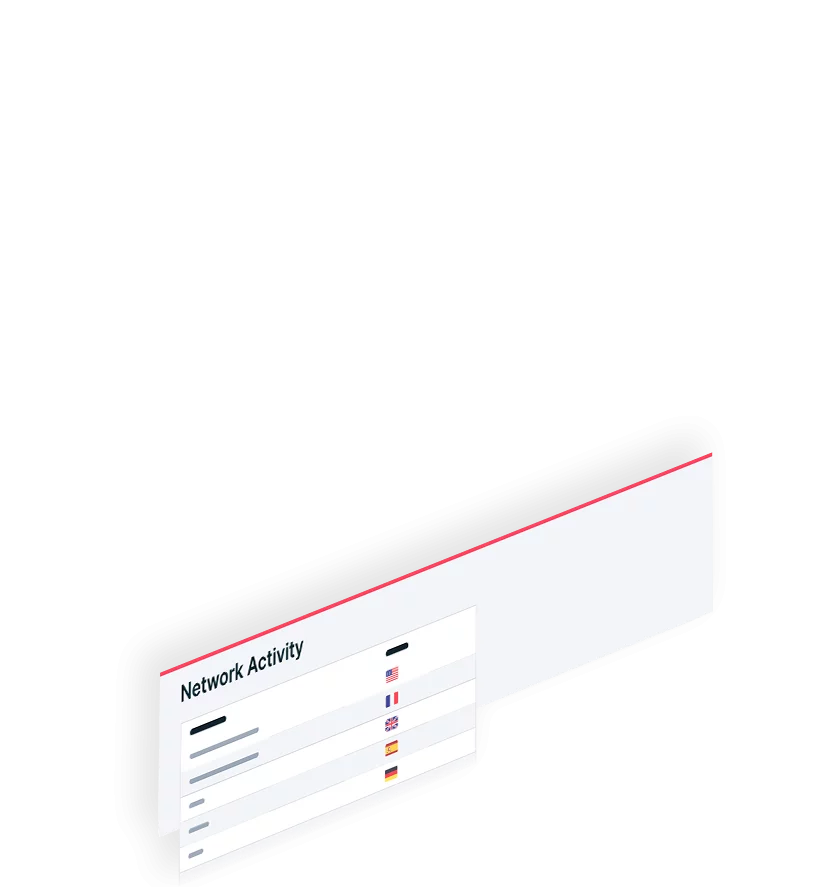
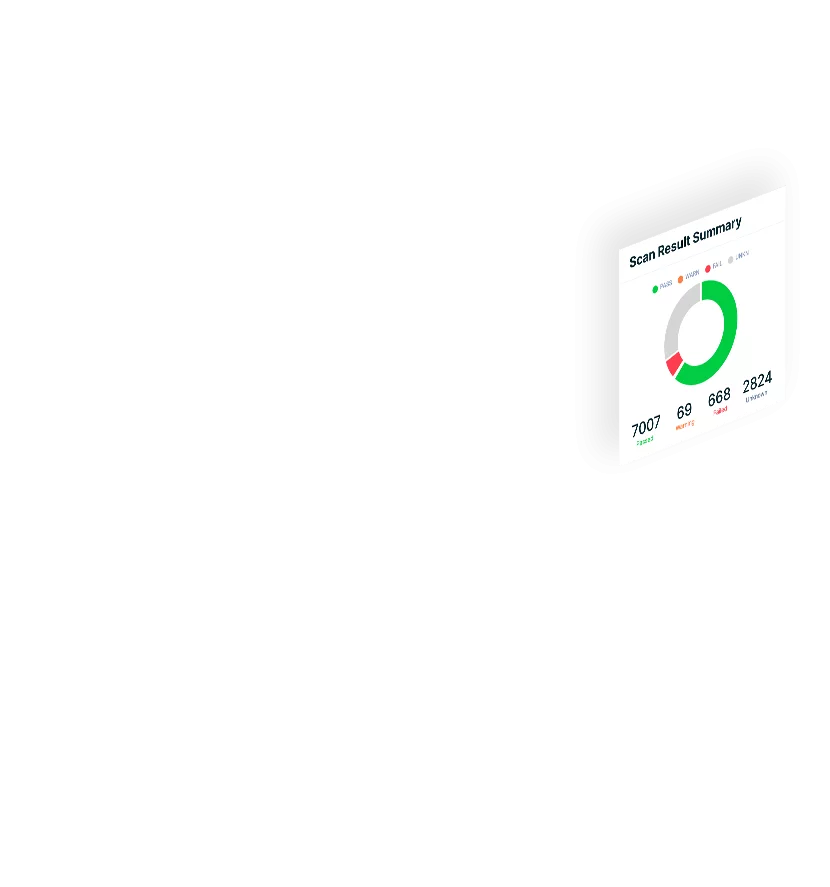
Quickly see and prioritize your cloud security risks to mitigate the most critical threats to your organization, stopping attacks in real time
One Platform
Bring teams together around the most comprehensive, fully integrated Cloud Native Application Protection Platform (CNAPP) to regain visibility and reduce risk to your business













幸运飞行艇开奖结果记录【官方】168体彩网 幸运飞行艇开奖历史记录查询 168飞艇计划软件官网直播 The freedom to run anywhere
Aqua secures your applications wherever you develop and run them
Across clouds, container and serverless platforms, CI/CD pipelines, registries, DevOps tools and modes of deployment, orchestrators, all the way to Security, SIEM, and Analytics.
Clouds
CI/CD
Container & Serverless
Container Registry
Security Tools
Finance
Enterprise
"Your Go-To Security Guys! It's one easy platform which contains all the necessary results and insights in regards to current security threats a system may be facing."
Security Engineer
Software & Technology
Finance
NCR Attains Security & PCI Compliance For Its
Container-Based Applications
Energy
"What I like best about Aqua Security is its comprehensive approach to security, proactive threat intelligence, strong focus on compliance, exceptional customer support, and commitment to continuous improvement."
Stefan James, Koch
Driving Security Innovation with Open Source
With a dedicated open source engineering team, we fuel security innovation in the cloud native ecosystem. We openly share our knowledge and capabilities and actively contribute to the community. This keeps the industry advancing and our enterprise customers ahead of what’s next.
Explore our projects